Imagine tracking every product instantly—but gaps in regulations, data, and supply chains make it chaotic.
Industries must overcome these challenges to ensure seamless regulatory traceability.
How Product Traceability Supports Compliance, Safety, and Supply Chain Efficiency
Regulatory traceability is pivotal in ensuring compliance, enhancing safety, and optimizing operational efficiency across industries. As global supply chains grow increasingly complex, the ability to track and document product movements is essential for risk management, fraud prevention, and regulatory adherence. However, implementing traceability standards has its challenges, from interoperability issues to high implementation costs.
Sector-specific regulations add another layer of complexity. Industries such as pharmaceuticals, food and beverage, electronics, automotive, medical devices, and consumer goods face distinct hurdles.
Let’s take a look at what they are.
Traceability Implementation: Challenges & Solutions
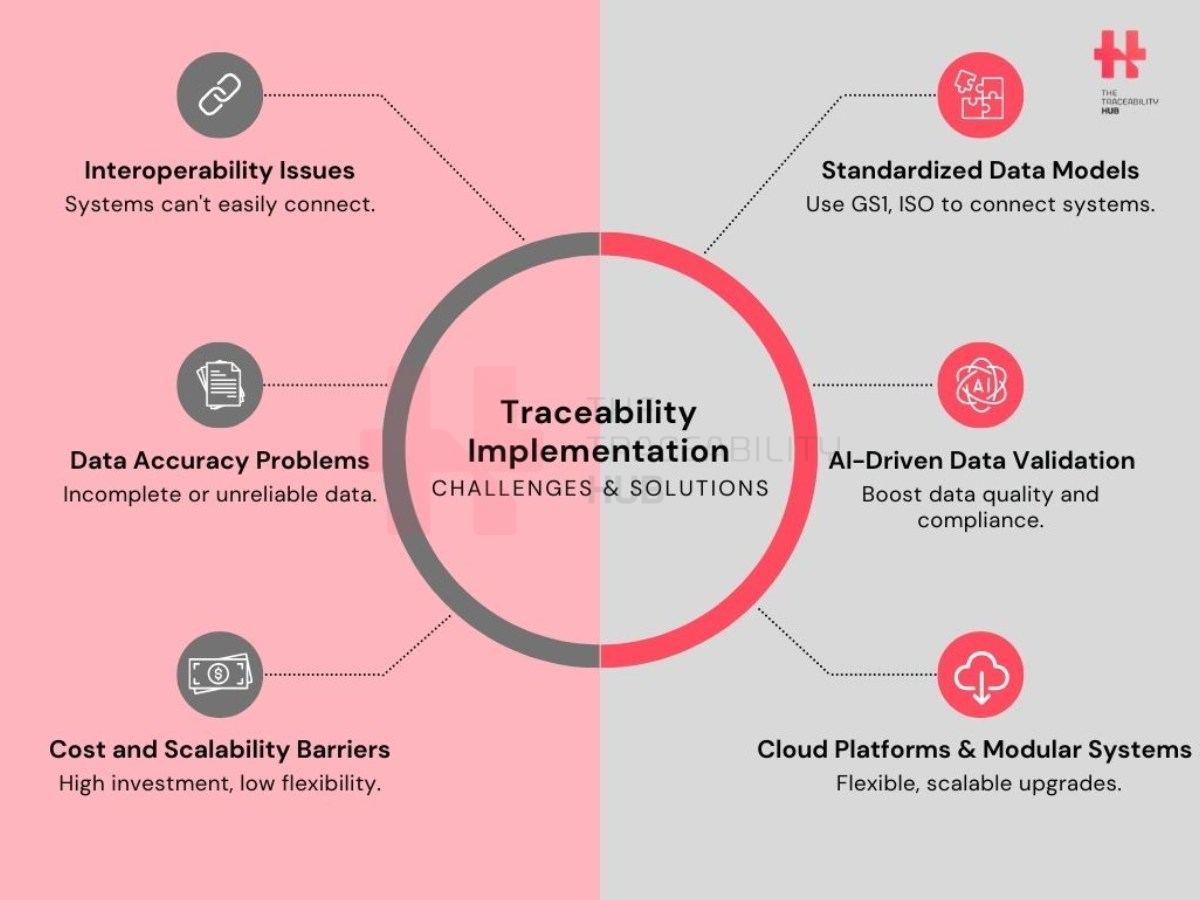
Common Challenges in Implementing Regulatory Traceability
Interoperability Issues
A key challenge is ensuring seamless integration between traceability systems and existing supply chain IT landscapes. Many organizations operate with legacy systems that lack compatibility with newer compliance platforms, creating data silos and inefficiencies.
Data Quality & Accuracy
Maintaining reliable traceability data is often underestimated: inaccurate or incomplete data can lead to compliance failures, increased recall risks, and inefficiencies in supply chain management.
Security & Compliance
Traceability systems must handle sensitive data while adhering to regulations. Implementing security measures to protect against cyber threats is essential.
Scalability & Cost Barriers
Regulatory changes necessitate continuous updates to traceability frameworks. However, modifying systems without incurring excessive costs remains a challenge, particularly for small and medium enterprises.
Change Management
Internal resistance and external dependencies, such as third-party logistics providers, can slow the adoption of new traceability solutions. Effective training and stakeholder engagement are vital for successful implementation.
Overcoming Traceability Barriers: Best Practices & Solutions
Gap Analysis & Compliance Audits
Organizations must identify regulatory gaps and ensure compliance with sector-specific mandates.
Standardized Data Models (GS1 EPCIS, ISO Standards)
Adopting universal data exchange standards simplifies interoperability and enhances supply chain transparency.
System Integration & IT Infrastructure
Cloud-based and API-driven solutions provide real-time compliance tracking, improving adaptability to regulatory updates.
Automation & AI-Driven Compliance Tools
Emerging technologies like artificial intelligence and blockchain streamline data validation, anomaly detection, and fraud prevention are important tools for compliance, cost efficiency, data gathering and business intelligence.
Change Management & Training Programs
Educating internal teams and supply chain partners ensures smoother adoption and adherence to compliance best practices.
Industry-Specific Challenges & Solutions
Pharmaceutical Industry
- Regulatory Frameworks: EU FMD, DSCSA (US), serialization & aggregation mandates.
- Challenges: Complex serialization requirements, data integrity, supply chain security.
- Solutions: Advanced coding & serialization, blockchain-based drug traceability.
Food & Beverage Industry
- Regulatory frameworks: FSMA (US), EU General Food Law, farm-to-fork traceability.
- Challenges: Contamination risks, fragmented supply chains, lack of real-time visibility.
- Solutions: Smart labelling (QR codes, RFID), digital product passports (DPPs).
Electronics Industry
- Regulatory frameworks: RoHS, WEEE directive, counterfeit prevention laws.
- Challenges: E-waste tracking, supplier verification, counterfeit risks.
- Solutions: Digital passports for electronics, AI-driven component verification.
Automotive Industry
- Regulatory frameworks: Battery passport (EU), ISO 26262, ELV directive.
- Challenges: Component traceability, end-of-life recycling mandates, sustainability compliance.
- Solutions: Battery passports, blockchain for component authentication.
Medical Devices Industry
- Regulatory frameworks: EU MDR, US FDA QSR, UDI (Unique Device Identification).
- Challenges: Strict safety standards, high serialization complexity, real-time tracking.
- Solutions: Unique device identifiers (UDI), cloud-based compliance systems.
Consumer Goods Industry
- Regulatory frameworks: CPSIA (US), GPSD (EU), eco-labelling laws.
- Challenges: Product recalls, labelling accuracy, sustainability reporting.
- Solutions: Smart packaging, AI-powered traceability solutions.
The Role of Traceability Platforms & Interoperability
Centralized traceability platforms enhance regulatory compliance by consolidating data across multi-tiered supply chains in one place. Interoperability remains a key consideration.
For this to work, collaboration among industry players to establish unified frameworks is crucial. Companies that have successfully integrated traceability solutions, such as through blockchain and cloud-based tracking, have demonstrated improved compliance and operational efficiency. Also the consulting company Gartner is mentioning the “Digital integration hub” as a key digital enabler, an architectural pattern that provides a scalable, and real-time layer for modern digital applications, while it centralizes data from various sources, and it aggregates data from multiple systems.
A platform well designed is making it easier to integrate and manage new data sources and applications without disrupting existing systems.
Future Trends in Regulatory Traceability Compliance
- AI, blockchain, and IoT adoption: Advanced analytics and distributed ledgers will further enhance traceability and fraud detection.
- Stricter global enforcement: Governments will impose tougher penalties for non-compliance, making adherence critical.
- Expansion of digital product passports (DPPs): More industries will adopt DPPs to streamline product authentication and lifecycle tracking.
Key Takeaways and Next Steps for Improving Supply Chain Traceability
Addressing traceability challenges requires a combination of standardized data models, scalable technology solutions, and robust change management strategies. As regulatory requirements evolve, organizations must continuously adapt to maintain compliance and operational efficiency.
Discover how implementing regulatory traceability standards can drive efficiency, reduce operational risks, and enhance compliance across industries. Learn about the key ROI factors and challenges businesses face when integrating these solutions.
Read more: The Cost-Benefit of Adopting Regulatory Traceability Standards: ROI & Challenges Per Industry






