Digital Leaflets & ePI for EU 2026 Compliance
Digital leaflets, also known as electronic Product Information (ePI) systems, are transforming how pharmaceutical companies manage regulatory information and patient communication. The revision of the EU General Pharmaceutical Legislation was proposed by the European Commission in April 2023 as part of the most comprehensive reform of EU pharmaceutical rules in over two decades. The legislative process is still ongoing, and while no fixed implementation date has been confirmed, digital product information is expected to play a central role in the modernization of regulatory frameworks across EU healthcare ecosystems.
It is important to clarify that electronic leaflets are distinct from the EU Falsified Medicines Directive (FMD). The FMD focuses on serialization and authentication requirements through unique identifiers and anti-tampering devices. Electronic Product Information (ePI), by contrast, addresses the digital structuring, accessibility, and updating of medicinal product information rather than anti-counterfeiting measures.
Electronic leaflets are part of an ongoing regulatory initiative led by the European Medicines Agency (EMA) and the Heads of Medicines Agencies (HMA). The goal is to develop a standardized, structured, and machine-readable format for product information, including the Summary of Product Characteristics (SmPC), package leaflets, and labeling. Structured digital medicine information enables faster updates to safety guidance and dosage recommendations while improving data consistency and cross-border accessibility.
The transition toward digital product information aligns with the broader revision of the EU pharmaceutical framework. An industry-led Inter-Association Task Force has proposed a phased implementation roadmap to support adoption. In parallel, the EMA/HMA electronic Product Information initiative is developing a centralized infrastructure and common standard intended to serve as a harmonized access point for structured product information across Member States.
Beyond the European Union, regulatory authorities in jurisdictions such as Japan, Singapore, and Brazil have introduced or expanded guidance supporting electronic labeling for medicinal products. This reflects a broader global shift toward digital pharmaceutical information systems designed to enhance regulatory agility, transparency, and patient access to updated medicine information.
Why Digital Leaflets Are Replacing Paper Inserts
The pharmaceutical sector is progressively integrating electronic Product Information (ePI) systems and digital patient information platforms to complement traditional paper inserts. While paper leaflets remain mandatory in many jurisdictions, regulatory modernization efforts increasingly support structured digital formats. This transition offers potential benefits for patients, healthcare professionals, and supply chain efficiency, while aligning with broader regulatory data standardization initiatives.
Instant Access Via QR Codes on Packaging
QR codes placed on medication packaging can provide direct access to digital product information hosted on regulatory-approved or manufacturer-controlled platforms. In certain Member States, such as the Netherlands, pilot frameworks have required that QR codes be accompanied by a visible URL to ensure accessibility for individuals who cannot scan codes.
Through a smartphone scan, patients and healthcare professionals may access up-to-date information including dosage instructions, contraindications, safety guidance, and interaction warnings. Digital access can be particularly useful in telehealth settings, where healthcare providers need remote access to structured product information.
Multilingual and Interactive Content for Better Understanding
Electronic leaflets improve accessibility by supporting multiple language versions, adjustable font sizes, searchable text, and compatibility with assistive technologies. Structured digital formats allow users to locate specific information more efficiently than navigating lengthy printed inserts.
Depending on implementation, digital platforms may also support enhanced accessibility features such as screen-reader compatibility or audio-enabled content. However, multimedia and interactive elements are subject to regulatory approval and are not universally implemented across all markets.
Real-Time Updates for Safety and Dosage Changes
One of the principal advantages of digital product information is the ability to update centrally hosted content when safety guidance or dosage recommendations change. Unlike paper inserts, which require reprinting and redistribution when revised, digital systems can reflect approved updates more rapidly once authorized by regulatory authorities.
QR codes linked to centralized ePI platforms can therefore provide access to the most recent approved version of product information. However, automatic patient alert systems are not inherent to QR codes themselves and depend on additional digital infrastructure.
Environmental Benefits: Reduced Paper Waste
Traditional paper leaflets require printing, packaging integration, and redistribution whenever updates occur, contributing to material consumption and associated logistics emissions across the pharmaceutical supply chain.
The adoption of electronic Product Information systems has the potential to reduce paper use and related environmental impacts. While precise environmental savings depend on regulatory implementation models and national requirements, lifecycle assessments generally indicate that digital information systems can lower material and distribution footprints compared to fully paper-based approaches.
As pharmaceutical regulation continues to evolve, structured ePI initiatives align with broader digitalization and sustainability objectives by improving information efficiency while reducing reliance on physical documentation.
Digital Leaflets & ePI
How Digital Leaflets Support EU 2026 Compliance
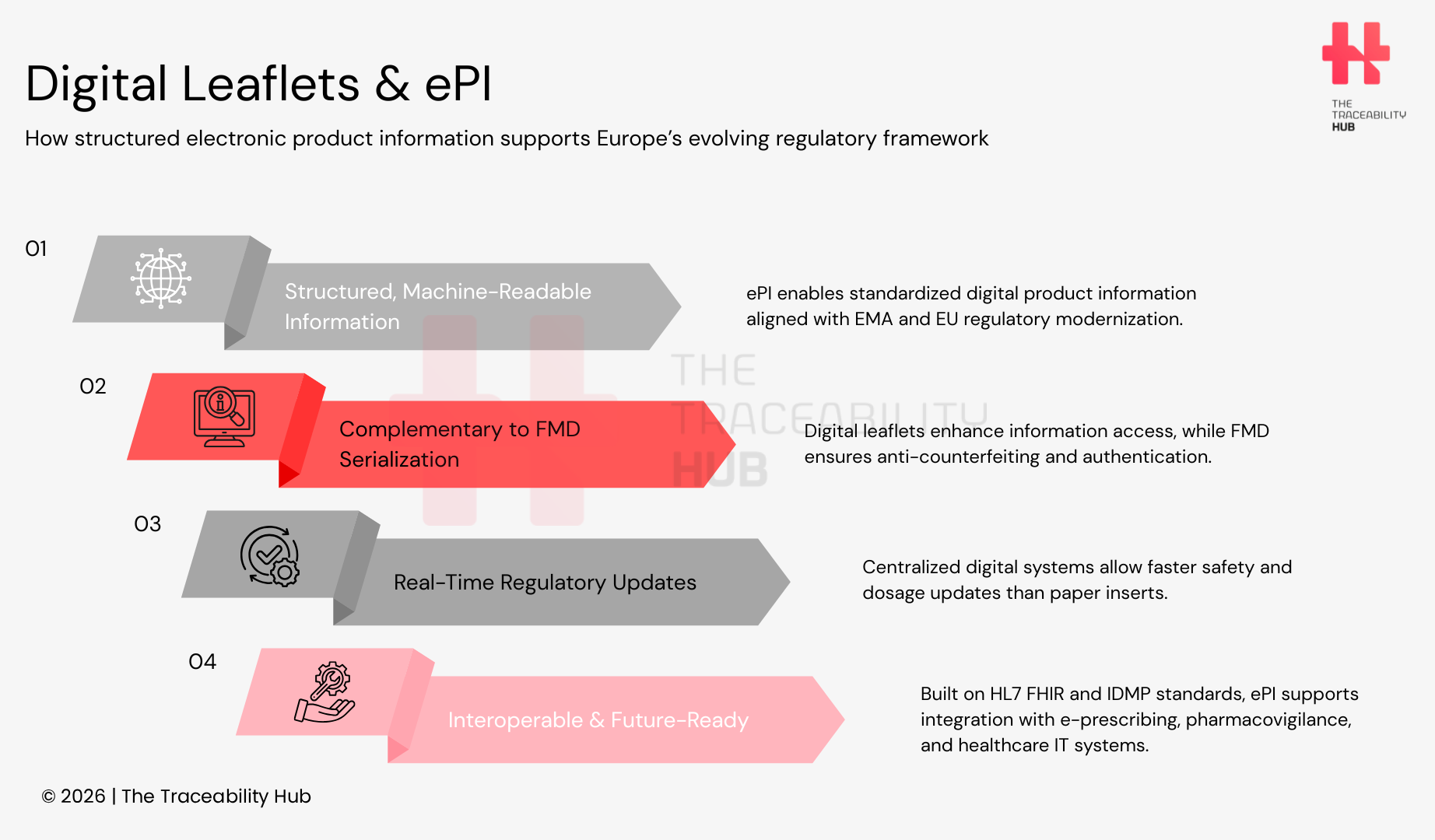
EU rules for medicines are evolving toward more digital, structured regulatory information, and electronic Product Information (ePI) is increasingly positioned as a key enabler of that shift. While ePI is not yet a blanket EU-wide legal requirement, EMA/HMA workstreams and the ongoing revision of the EU General Pharmaceutical Legislation indicate a clear direction toward standardized digital product information to improve patient access, update speed, and data interoperability.
EU Falsified Medicines Directive (FMD) Serialization Requirements
The EU Falsified Medicines Directive introduced safety features to protect the legal supply chain from falsified medicines. In practice, the safety-features system (set out in Commission Delegated Regulation (EU) 2016/161) requires a unique identifier (2D DataMatrix) and an anti-tampering device on the outer packaging of most prescription medicines, and certain non-prescription medicines where required by risk assessment. These requirements have applied across the EU since 9 February 2019, with authentication performed at dispensing.
Digital leaflets are not required by the FMD, but they can complement serialization strategies when packaging carries a scannable code that links users to structured, regulator-approved product information (e.g., via an ePI page). This supports access and transparency, while remaining distinct from anti-counterfeiting verification (which is handled through the FMD repositories system).
EMA ePI Pilot and Structured Content Mandates
Electronic product information (ePI) is an initiative led by the European Medicines Agency (EMA) together with the Heads of Medicines Agencies (HMA) to enable structured, machine-readable product information based on the EU ePI Common Standard. From July 2023 to August 2024, EMA and national competent authorities in Denmark, the Netherlands, Spain, and Sweden ran an ePI pilot in live regulatory procedures. The pilot resulted in the publication of 23 ePIs (covering medicines handled through EMA and national procedures) and generated practical recommendations on how to integrate ePI into regulatory workflows and expand its use.
Print-on-Demand Safeguards for Inclusion
A key challenge in moving from paper leaflets to digital access is digital inclusion. Eurostat reported that 56% of people in the EU aged 16–74 had at least basic digital skills in 2023, underlining the need for safeguards for people who may not be able to access information digitally.
In this context, the industry-led Inter-Association Task Force (IATF) on ePI (AESGP, EFPIA, Medicines for Europe) has proposed a phased approach to ePI adoption and discussed complementary measures to ensure continued access to paper information, including approaches commonly referred to as “print on demand” during transition scenarios. At the same time, stakeholders have raised operational concerns around print-on-demand models (workload, cost, workflow impacts), which remain part of the policy debate as the EU’s pharmaceutical legislation revision progresses.
Alignment with IDMP and Regulatory Data Standards
To function as a modern regulatory data layer, digital product information needs to align with established data standards, particularly ISO IDMP (Identification of Medicinal Products), which is designed to enable consistent identification and exchange of medicinal product data across regulators and healthcare systems. EMA’s IDMP program supports interoperability across regulatory and healthcare communities, improving data consistency for cross-border use cases.
At EU level, Commission Implementing Regulation (EU) No 520/2012 (pharmacovigilance) references ISO IDMP standards and sets related obligations (including in Articles 25 and 26) that underpin structured medicinal product data exchange. In practice, alignment between ePI and IDMP supports interoperability with e-prescribing, pharmacovigilance systems, and electronic health records by anchoring product information to standardized identifiers and structured datasets.
Implementation of ePI Across EU Member States
Implementation of electronic Product Information (ePI) across EU Member States is progressing at different speeds, reflecting differences in national digital infrastructure, regulatory priorities, and the maturity of local product-information systems. The ePI initiative is coordinated by the European Medicines Agency (EMA) together with the Heads of Medicines Agencies (HMA), and participation has so far been driven through pilots and technical collaboration rather than a single uniform rollout across all Member States.
The pace of adoption is expected to depend on national readiness, legislative alignment at EU level, and the ability to integrate structured ePI into existing regulatory databases and national distribution channels, which may result in differentiated timelines across the EU.
Centralized Access Via EMA/HMA Portal
The EMA Product Lifecycle Management (PLM) portal is already used as part of the ePI pilot ecosystem, enabling participants to create, submit, and publish ePI in structured formats and to make published pilot ePIs accessible via a dedicated interface and API. At present, access to authoring functions is limited to pilot participants, and published ePIs are clearly marked as pilot outputs.
Looking ahead, the PLM portal and associated services are widely viewed as an enabling platform for scaled ePI implementation, but the EU-wide operating model—including whether and how it becomes a comprehensive access point across Member States—will depend on final legislative decisions and implementation governance.
Country-Specific Timelines and Hosting Models
Member States are exploring different approaches to digital product information, including pilots focused on hospital medicines and cross-border availability. In the Nordic region, regulatory cooperation has tested English-language common Nordic packs for selected hospital medicines, supported by digital access to product information in national languages, as a model to improve availability while maintaining local-language information access.
Belgium and Luxembourg have also run hospital-focused initiatives assessing the feasibility of removing paper package leaflets for medicines restricted to hospital use, replacing them with digital access to information through trusted sources in controlled healthcare settings. Evidence from these pilots indicates strong acceptance among hospital professionals, while broader EU-wide adoption remains subject to policy and legislative evaluation.
QR Code vs GS1 DataMatrix Usage by Region
Across Europe, the technical identifiers used on medicine packs serve different purposes. Under the EU Falsified Medicines framework, most prescription medicines carry a 2D DataMatrix unique identifier for verification in the supply chain, while QR codes—where used—typically support information access rather than authentication. Over-the-counter (OTC) medicines generally fall outside the mandatory unique identifier requirement (with exceptions where specific products are designated), so digital leaflet access for OTC products may rely on alternative scannable codes and national implementation choices.
Transition Strategies for HCP-Administered vs OTC Products
Proposed transition approaches often differentiate between medicines administered by healthcare professionals in controlled settings and products used directly by patients. Industry groups have argued that hospital- and HCP-administered products could transition earlier to digital-first leaflet access following legislative enactment, while self-administered medicines may require more gradual change to ensure inclusion, continuity of access, and alignment with national healthcare pathways. This staged logic is reflected in industry position work and in the way several EU pilots have been structured to start with hospital settings before broader expansion.
Future of Digital Leaflets in the EU Ecosystem
Digital leaflets, delivered through electronic Product Information (ePI), are evolving as part of the EU’s broader shift toward structured, interoperable regulatory data. Rather than changing medicine verification systems themselves, ePI initiatives focus on improving how approved product information is structured, maintained, and accessed by patients and healthcare professionals, with the goal of enabling faster updates, better consistency, and improved digital accessibility across the European healthcare ecosystem.
HL7 FHIR-Based Structured ePI Adoption
The European Medicines Regulatory Network has published a FHIR Implementation Guide that specifies how HL7 FHIR resources can be used to create ePI in line with the EU ePI Common Standard. This confirms the technical direction of travel toward structured, machine-readable medicine information that can be exchanged consistently across systems and Member States.
This technical foundation is intended to support future scaling of ePI within regulatory processes, enabling more consistent creation and exchange of product information while maintaining alignment with EU governance and regulatory workflows.
In parallel, the Gravitate Health consortium has collaborated with EMA and a wider international community to support convergence toward a globally harmonised approach to structured ePI using HL7 FHIR resources, including engagement activities with regulators beyond Europe.
AI-Powered Personalization of Patient Information
Artificial intelligence is increasingly being explored to improve how patients understand health information by adapting language, presentation, and navigation to different literacy levels and needs. While evidence on “AI personalization” specifically is still emerging, research on digital health interventions and tailored communication shows that technology-enabled approaches can improve medication adherence in some contexts, although outcomes vary significantly by condition, population, and implementation model.
Any use of AI in patient-facing medicine information must operate within strict regulatory, clinical governance, and data-protection constraints—particularly where sensitive health data is involved—and should be designed to avoid introducing uncontrolled variations in regulated content.
Blockchain for Leaflet Authenticity and Traceability
Blockchain has been explored in the pharmaceutical sector to strengthen data integrity, improve traceability, and support product-information trust models. This sits alongside—rather than replacing—the EU’s official verification framework under the Falsified Medicines Directive, which relies on serialization, anti-tampering features, and repository-based verification across the supply chain.
The World Health Organization has highlighted that substandard and falsified medicines remain a significant public health issue, particularly in low- and middle-income countries.
Within EU-funded innovation, projects such as PharmaLedger (supported under the Innovative Medicines Initiative / successor programs) have piloted blockchain-enabled approaches to product verification and trusted data exchange, including concepts for linking packs to authoritative electronic product information. These pilots illustrate one possible “trust layer” model but remain largely experimental and must be evaluated against cost, scalability, and governance requirements.
Voice-Enabled ePI for Accessibility
Directive (EU) 2019/882 (the European Accessibility Act) establishes accessibility requirements for a range of products and services, reinforcing the EU’s broader policy direction toward inclusive digital environments. The European Commission estimates that around 87 million people in the EU live with some form of disability, underscoring the importance of accessible digital health information.
As ePI systems evolve, accessibility considerations—such as screen-reader compatibility, adaptable typography, and text-to-speech support—become increasingly relevant to ensure equal access to regulated medicine information. While voice features are not mandated specifically by the ePI framework, accessibility-by-design aligns with the EU’s wider objectives for inclusive access to essential information in the digital age.
Digital Leaflets Meet EU 2026 Compliance
Digital leaflets and structured ePI are increasingly positioned as part of Europe’s transition toward digital regulatory information ecosystems, especially as EU policy and national implementations continue to advance. However, there is no single EU-wide “2026 deadline” that mandates replacement of paper leaflets with ePI across all medicines. Instead, progress is occurring through pilots, technical standards, and phased policy discussions focused on improving access, update speed, and interoperability of approved product information.
Where digital access is implemented, patients can retrieve product information through scannable identifiers on packaging, with potential benefits such as multilingual access, improved navigation, and faster availability of updated, authority-approved content. Healthcare professionals can also benefit from easier remote access to consistent product information. At the same time, any transition must address inclusion and continuity of access, ensuring that patients who cannot use digital channels still receive the information they need.
From a technology perspective, HL7 FHIR-based standards provide the foundation for integration between ePI and healthcare IT systems, supporting interoperability across regulatory and clinical contexts. AI and advanced analytics may further enhance how information is presented and understood, but must be introduced within strict governance, privacy, and regulatory controls. Parallel innovation efforts—such as blockchain pilots—illustrate how trust and integrity layers might evolve, even though the EU’s official verification framework remains repository-based under the Falsified Medicines system.
Overall, the shift toward digital medicine information has the potential to improve access and regulatory agility while supporting sustainability objectives by reducing reliance on reprinted paper documentation. Its long-term success will depend on consistent standards, strong governance, and inclusive implementation models that protect patient safety and maintain equitable access to essential product information.
Read more: Proven Serialization Methods to Secure Alcohol Supply Chains






