Why This Matters: Regulation as a Market Signal, Not Just a Constraint
For international beauty brands, Russia is a market of scale, sophistication, and appetite for high-quality cosmetic products. But the rules of access are changing fast. What was once a matter of certificates, labels, and batch documentation is evolving into something far more structural: full digital traceability at unit level.
With the expansion of the Chestny ZNAK (Честный ЗНАК), which translates to “Honest Sign” or “Honest Label” in Russian, is the national product traceability and labeling system in Russia, for cosmetics it is starting in 2025, making the regulatory compliance no longer a background regulatory task. It becomes a core operational capability, one that directly affects market access, supply chain visibility performance, brand protection, and consumer trust.
At its core, the regulation is designed to address three systemic goals:
- Fight counterfeiting and grey market imports, which have long distorted competition and eroded brand value and integrity.
- Protect consumers by ensuring that every product on the shelf is authentic, safe, and traceable through its entire product genealogy.
- Create transparency and real-time market oversight, giving authorities visibility into product flows from production to the final point of sale.
The practical consequence of this approach is profound. Russia is building a closed-loop digital ecosystem in which a single Data Matrix code, acting as a unique digital identifier, follows each product throughout its lifecycle. That same code simultaneously serves regulators, logistics operators, retailers, brands, and even consumers, who can independently verify authenticity through a mobile app.
For brands, this means one thing above all: regulatory compliance is unavoidable. There is no alternative route to the market.
But there is a decisive difference between being compliant and being well implemented. Companies that treat Chestny ZNAK as a minimal obligation risk operational friction, delays, and escalating costs. Those that approach it as a structured transformation, integrated into packaging, IT, and supply chain processes, can turn regulatory pressure into a competitive advantage.
In this new context, how compliance is designed, executed, and scaled matters just as much as compliance itself.
Timeline & Scope: What Changes, When and What’s In
Chestny ZNAK is Russia’s national digital product marking and traceability platform infrastructure. Established through federal legislation in 2017, the system has progressively expanded from pilot programs into mandatory digital labeling across multiple regulated product categories:
- Food and beverages
- Dairy products (milk, cheese, yogurt, butter, fermented products)
- Bottled water (mineral and drinking water)
- Canned foods (fish, vegetables, fruit, etc.)
- Caviar and fish roe
- Snacks, sauces, and spices
- Vegetable oils (sunflower, olive, rapeseed, etc.)
- Alcoholic beverages
- Beer and beer-based beverages
- Spirits
- Tobacco products
- Cigarettes
- Pharmaceuticals & health products
- Medicines (including over-the-counter products)
- Biologically active supplements / dietary supplements
- Apparel & textile products
- Clothing and textiles (including light industrial textile products and fur)
- Footwear
- Consumer goods & industrial products
- Tires and tire covers
- Perfumes and cosmetics (including cologne)
- Cameras and flashes
- Electronics (smartphones and selected appliances)
Perfumes and eau de toilette, for example, have been subject to mandatory digital marking unit-level serialization since 2020.
The cosmetics sector is now entering a phased expansion stage under the national rollout plan.
2025 Cosmetics Rollout (Phased Implementation)
According to the approved regulatory roadmap, mandatory digital labeling for cosmetics is expected to expand in stages during 2025, including:
- From 1 March 2025: Mandatory registration (not marking).
- From 1 May 2025: Soaps / detergents marking begins.
- From July 2025: Deodorants, hair care, and shaving products.
- From October 2025: Cosmetics and toothpaste.
Each implementation milestone activates operational requirements across the supply chain. Once a category becomes subject to mandatory marking, products must carry the required marking codes (means of identification) and be introduced into circulation in Chestny ZNAK according to the rules for that product group.
This affects:
- Packaging design and printing
- Serialization infrastructure and API-based integration for data flow
- Production and aggregation processes
- Import and customs procedures and due diligence documentation
- Warehouse and retail scanning systems for real-time data capture
Companies operating in affected categories must align production and logistics timelines with regulatory activation dates to avoid disruption of sales and maintain operational resilience within the Russian Federation.
The Longer Arc: from Category Rollout to Full Traceability
The expansion of digital labeling for cosmetics and household chemicals under Chestny ZNAK follows a structured, multi-year regulatory pathway rather than a one-time switch. After initial category activation in 2025, the system transitions through staged accounting models before reaching full item-level traceability.
For cosmetics and household chemicals, regulations provide for:
- Progressive mandatory marking beginning in 2025 for defined product groups.
- A volume-sort accounting phase from 1 July 2026 to 30 June 2028, during which reporting is based on batch/volume-level documentation rather than individual code reconciliation.
- Transition to full item-by-item accounting from 1 July 2028, requiring detailed reporting of individual unique product codes across supply chain events.
This means compliance maturity increases over time: from basic unit-level marking to comprehensive serialization management and audit trails across production, import, warehousing, and retail.
What about Existing Inventory?
Transitional rules apply to products manufactured or imported before the mandatory marking date for a given category. In the cosmetics and household chemicals group, official guidance indicates that, where applicable, previously released unlabeled goods may be sold until the end of their shelf life, provided they were legally placed on the market before the enforcement date.
However, sell-through allowances do not eliminate operational risks. Cosmetics companies must prevent mixing compliant and non-compliant stock, ensure correct warehouse segregation, and leverage multi-tier supplier mapping to avoid shipping goods that will fall out of compliance once mandatory marking becomes effective for that category.
What Products Are We Talking About?
The cosmetics and household chemicals rollout cover a broad commercial spectrum. Based on official category guidance, included product groups expand in stages and may include:
- Soaps and detergents
- Hair care products (e.g., shampoo, conditioners, styling products)
- Deodorants and shaving products
- Decorative cosmetics and skincare products
- Toothpaste and oral care products
- Household chemical products such as cleaning agents
Final inclusion is determined by official product classification codes (TN VED / OKPD2), meaning companies must verify whether specific SKUs fall within scope.
In practical terms, most mainstream beauty, personal care, and household-care products placed on the Russian market will be subject to digital marking either during the 2025 rollout phase or during subsequent expansion toward full item-level traceability by mid-2028.
Beauty Exports to Russia
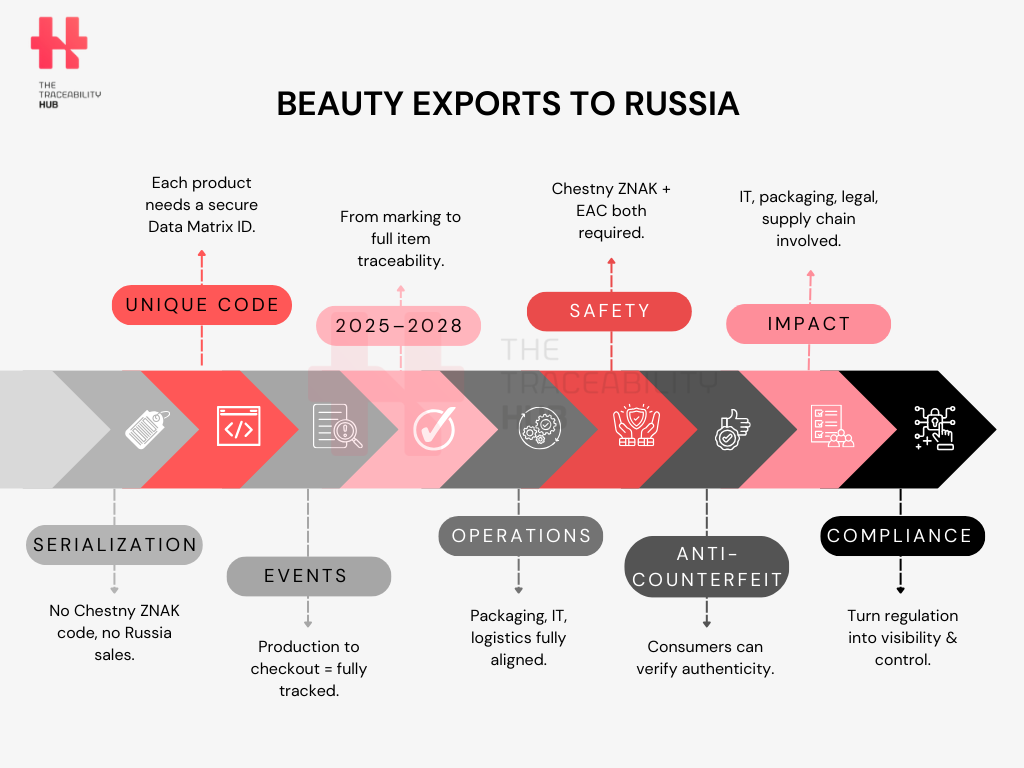
What Chestny ZNAK Actually Requires: Codes, Events, and Proof at Checkout
To many beauty companies, “digital labeling” can sound like a packaging tweak, add a code, update artwork, and move on. Chestny ZNAK is different. It’s not just a label. It’s a system of identity and reporting that follows each product from production (or import) all the way to the retail checkout.
Think of it as Russia’s version of a product “passport” (similar to the ESPR-mandated Digital Product Passport) plus a “travel log”, and both must be accurate, complete, and aligned.
The Code: A Digital Passport for Every Single Unit
Under Chestny ZNAK, every individual sales unit requires a 2D Data Matrix code. This is the foundation of the entire traceability model, because it makes each item uniquely identifiable, not just the batch or the shipment.
Each code typically includes:
- GTIN (often GTIN-14 in Russia)
- A randomized serial number as a unique identifier
- A cryptographic signature designed to prevent cloning or manipulation and ensure brand integrity
- Optional fields such as batch number and expiry date, when used by the brand/category
This means compliance is not achieved by “marking the box.” It is achieved by creating a unique, verifiable identity for every unit placed on the market.
Event Reporting: the Lifecycle Must Be Digitally Recorded
The second pillar is often the one that catches companies off guard: it’s not enough to print the code. Market participants must report events to the central traceability system as products move through the supply chain.
Key actors typically include:
- Manufacturer / importer (the party responsible for requesting codes and putting marked goods into circulation)
- Distributors / wholesalers (handling movements and transfers)
- Retailers (final scan at sale)
Typical events to report include:
- Code request and issuance
- Printing / application at the packaging line
- Aggregation (digital linking of unit → case → pallet)
- Shipment / receiving and relevant ownership or custody changes (ensuring end-to-end visibility)
- Retail scan at checkout, which officially marks the item as withdrawn from circulation
In other words: Chestny ZNAK is built on the logic that a code without events is incomplete, and events without reliable codes are unusable.
Consumer Verification: the Buyer Becomes Part of the Control Loop
What makes this system especially powerful is that traceability is not only for authorities and supply chain operators. Consumers can scan products using the official Chestny ZNAK mobile app to confirm authenticity and legal market status.
This creates a closed-loop control model in which a single unit-level product identity supports regulatory oversight, enables authorities to monitor introduction and circulation events, reduces the risk of counterfeit distribution, enhances supply chain reporting accuracy as traceability requirements mature, and allows consumers to verify the validity and legal market status of products through the official Chestny ZNAK application.
Chestny ZNAK is compliance-by-design. The brands that implement it as an end-to-end capability (code integrity + data integrity + operational reporting) are the ones that stay fast, avoid disruption, and position themselves to extract business value beyond the regulation.
The Hidden Complexity Behind “Just Adding a Code”
On paper, Chestny ZNAK looks straightforward: apply a Data Matrix, report events, stay compliant. For most cosmetics companies, this is the first real encounter with full unit-level serialization, and that’s where complexity quietly accumulates.
What often slows projects down is not the regulation itself, but the number of interdependent operational changes triggers across packaging, IT, logistics, and broader organization.
Packaging & Printing: Where Compliance Meets Brand Equity
The first friction point is physical. Every unit needs a scannable, high-quality code, and that code must live on packaging that was never designed for it.
Common challenges include:
- Redesigning artwork to create space for the Data Matrix on the label or the product, without compromising aesthetics or brand hierarchy
- Maintaining print quality on demanding substrates such as:
- Lacquered cartons and metallic foils
- Aerosols, tubes, sachets, doypacks, and other flexible formats
- Ensuring consistent readability at speed, which requires vision inspection systems and automated rejection handling for non-compliant units
For beauty brands, where packaging is a core part of perceived value, this balance between regulatory compliance and visual identity is non-trivial.
Aggregation Realities: Part No One Can Skip
While not always intuitive at the start, aggregation quickly becomes non-negotiable, both for regulatory compliance and operational efficiency.
Aggregation links individual serialized units to cases and pallets, enabling:
- Faster warehouse operations and real-time data capture
- Compliant shipment reporting and end-to-end supply chain visibility
- Manageable recalls management and inventory control
The hardest scenarios tend to be:
- Multipacks and bundles
- Promotional kits and gift sets
- Mixed-SKU packaging that changes seasonally
Without robust aggregation of logic, reporting becomes error-prone, and logistics slows down, exactly what brands want to avoid.
IT and Data Integration: The Real Backbone of the System
Chestny ZNAK is, at its core, a data-driven ecosystem. That means serialization cannot sit in isolation.
Typical required integrations include:
- ERP and MES, where product and production master data originate
- Warehouse Management Systems (WMS) for inventory and movements
- Logistics partners, often via EDI or similar data exchanges
- Direct API-based integration and connectivity with Chestny ZNAK / CRPT interfaces
On top of connectivity, there is governance:
- Master data accuracy (GTINs, product descriptions, hierarchies)
- Ensuring serialization data integrity
- Audit trails and event logs that can withstand regulatory inspection
This is where fragmented, patchwork solutions tend to break down.
Organizational Impact: Compliance Becomes Cross-Functional
Finally, Chestny ZNAK rarely belongs to a single department. Successful implementations almost always involve coordinated effort across:
- Packaging and artwork teams
- IT and data management
- Regulatory and legal affairs
- Supply chain and logistics
- Quality and operations
- Marketing (and Sales)
That’s why leading brands no longer treat Russian serialization as a marginal regulatory task. It’s a cross-functional transformation project, where early alignment determines whether compliance becomes a bottleneck, or a capability.
The “hidden complexity” is real, but predictable. Brands that acknowledge it early, design it holistically, and avoid siloed decisions are the ones that stay compliant without slowing down the business.
Don’t Forget EAC Conformity: Traceability Isn’t Safety Compliance
A frequent mistake among exporters is to treat Chestny ZNAK as “the Russia requirement.” It isn’t. Chestny ZNAK governs traceability and market circulation, but it does not replace the core product safety framework.
In other words: Chestny ZNAK ≠ product safety compliance.
To legally place cosmetics and make-up on the market, most brands must address two parallel obligations:
- EAC conformity assessment for safety (product compliance)
- Chestny ZNAK digital labeling (traceability regulatory compliance and circulation control)
The Baseline: EAC Declaration under TR CU 009/2011
For most cosmetic products sold in Russia and the Eurasian Economic Union (EAEU), the standard route is an EAC Declaration of Conformity under TR CU 009/2011 (“On the safety of perfumery and cosmetic products”). This declaration is a formal, legally binding statement that the product meets required safety standards, typically supported by accredited laboratory testing.
High-Risk Categories: When State Registration Applies
Some product categories go beyond the declaration and require state registration (often via competent authorities and accredited bodies). These are typically “higher risk” or more sensitive use cases, such as:
- Children’s cosmetics
- Intimate care cosmetics
- Skin whitening and artificial tanning products
- Chemical peels and depilatories
- Hair lighteners and chemical straightening/curling products
- Products involving nanomaterials
- Certain oral care products with higher fluoride thresholds
- Teeth whitening products containing hydrogen peroxide (or peroxide-releasing ingredients)
- Components for permanent make-up / tattooing
For brands with broad portfolios, this distinction matters: it can change timelines, documentation effort, and testing scope.
A Key Regulatory Update (Effective December 24, 2025)
Companies active in the EAEU should note that amendments to TR CU 009/2011 adopted by EEC Council Decision No. 114 of 29 November 2024 entered into force on 24 December 2025, with certain provisions applying later, including from 1 July 2026. The changes update core compliance elements such as terminology and definitions, scope and exclusions, conformity assessment procedures and documentation, and consumer packaging requirements (including new packaging integrity and presentation rules for specific product types).
As a result, compliance programs cannot be “set and forget”: they require active governance, version control, and audit-ready documentation, especially when reformulating products or expanding SKUs.
Testing: What EAC Conformity Typically Relies on
Before an EAC Declaration (and, where required, state registration) can be completed, products generally undergo rigorous testing such as:
- Physicochemical assessment
- Microbiological evaluation
- Toxicological assessment
- Clinical evaluation (for specific categories / claims)
To export cosmetics to Russia without disruption, brands must treat traceability and safety as two coordinated tracks. Chestny ZNAK protects the circulation of each unit through unit-level serialization. EAC conformity protects the legitimacy of the product itself. Both are required, and aligning them early is often the difference between a smooth launch and a costly delay.
How to Turn Compliance into Advantage
Chestny ZNAK (within the wider EAEU/EAC compliance environment) can initially feel like “just more regulation.” However, once marking, reporting, and data governance are operational, the same infrastructure can support stronger control mechanisms, more consistent operations, and consumer-facing verification.
Brand Protection
Anti-counterfeit controls are strengthened at item level through a unique marking code encoded in a DataMatrix and supported by verification elements (verification key and verification code, often referred to as a “crypto tail”), which makes simple replication significantly harder.
In addition, event-based reporting can improve accountability across importers, distributors, and retailers by creating clearer documentation of product circulation and movements through the chain.
Operations
Unit-level marking and structured reporting can improve visibility into what has been introduced into circulation and how goods move through the supply chain, especially where aggregation and standardized logistics processes are implemented. This foundation can also support more efficient investigations and corrective actions when specific units need to be identified, and it supports audit readiness by creating consistent digital records that reduce manual reconciliation during inspections.
Consumer & Marketing
A distinctive feature of Chestny ZNAK is that consumers can participate in verification. Using the official mobile app, buyers can scan marking codes to check whether a product is validated in the state system and view relevant product information, which can reinforce trust at the point of purchase. Brands may also choose to layer voluntary digital experiences on top of compliant marking (for example, how-to content or brand education), provided these do not conflict with mandatory labelling and regulatory requirements.
Strategic Readiness
Investments in serialization workflows, master data discipline, and integration patterns built for Chestny ZNAK can be repurposed as internal capabilities, especially as more markets adopt traceability and digital product identification requirements. Standardizing packaging rules, data governance, and integration can also reduce friction when expanding portfolios or introducing new SKUs.
Choosing the Right Partner: Why Integration Matters
Implementing Chestny ZNAK compliance is not a single technical adjustment: it is a complex orchestration of packaging, IT systems, regulatory processes, and real-time data exchange. Too often, companies attempt to “stitch together” separate vendors for printing, vision inspection, data management, ERP integration, and regulatory reporting. The result can be fragmented accountability, inconsistent performance, and increased operational risk.
In a system as strict and interconnected as Chestny ZNAK, gaps between suppliers quickly become vulnerabilities: unreadable codes, failed aggregation, reporting mismatches with CRPT, or incomplete audit trails can delay shipments, block customs clearance, or expose brands to penalties. The more fragmented the architecture, the higher the operational risk.
What a Technology Partner Must Cover
To ensure smooth and scalable regulatory compliance, a reliable partner should provide a structured, end-to-end framework covering:
- Line-level serialization and aggregation directly on cosmetic packaging lines
- High-quality printing and vision inspection systems to guarantee Data Matrix readability and compliance
- Secure connectivity and automated reporting to CRPT, minimizing manual intervention and error
- Compliance monitoring dashboards with full audit readiness and traceability logs
- Advanced data management tools to transform serialized data into operational and marketing insights
This is not simply about applying a code, it is about building a stable digital infrastructure that supports manufacturing, logistics, regulatory reporting, and brand strategy simultaneously.
Antares Vision Group: An Integrated Approach
This is where Antares Vision Group positions itself as a strategic partner rather than a component supplier.
Through an integrated platform, Antares Vision Group enables:
- Serialization and aggregation directly on cosmetic packaging lines
- Automated and secure connectivity with the Chestny ZNAK system
- Real-time compliance monitoring dashboards
- Data intelligence tools that allow brands to extract operational efficiency and marketing value from serialized data
Instead of coordinating multiple disconnected providers, brands can rely on a unified technological ecosystem that ensures continuity, accuracy, and regulatory security.
For companies entering the Russian market, or expanding their product portfolio, this integrated model significantly reduces implementation risk, shortens timelines, and transforms compliance from a burden into a structured competitive capability.
A Structural Shift, Not a Labelling Tweak
Chestny ZNAK is not a simple compliance add-on. For cosmetics brands, it represents a structural transformation in how products are identified, packaged, documented, shipped, and ultimately sold in Russia. Moving from batch-level controls to unit-level serialization with mandatory event reporting reshapes the entire operating model, touching packaging design, line performance, IT architecture, warehousing, retail execution, and data governance.
With the first key deadlines starting in 2025, preparation is no longer optional or “later.” Brands that treat this as a last-minute labelling project risk disruption: delayed launches, blocked shipments, non-compliant stock, and costly rework across artwork, packaging lines, and systems integration.
The strongest outcomes typically come from three factors:
- Starting early to secure timelines for packaging updates, onboarding, testing, go-live stability and system integration.
- Building strong data foundations, from GTIN structure and product master data to audit logs and end-to-end reporting integrity.
- Choosing an experienced end-to-end technology partner, capable of managing serialization, aggregation, print and vision quality, secure CRPT connectivity, and compliance monitoring in one integrated framework.
In short: brands that invest now won’t just protect their Russian market access—they’ll build traceability capabilities that improve control, operational resilience, and competitiveness well beyond compliance.
Read more: Traceability Platforms: Enabling Compliance and Supply Chain Visibility






