Verified Overview: GCC Regulatory Affairs Summit 2026
The GCC Regulatory Affairs Summit 2026 is a major regional conference focused on pharmaceutical regulatory affairs, compliance frameworks, GCC compliance frameworks, regulatory harmonization initiatives and policy developments across the Gulf Cooperation Council (GCC) markets. Scheduled for 20–24 April 2026 at the Mövenpick Grand Al Bustan in Dubai, UAE, the summit features multiple flagship events, including the Regulatory Affairs Pharma Summit, Pharmacovigilance Training, GCC Quality Conference and eCTD Training, designed to support regulatory professionals with practical insights and knowledge exchange.
This multi-day platform brings together regulators, industry representatives, compliance experts and solution providers from across the GCC to explore regulatory frameworks GCC regulatory frameworks, pharmaceutical harmonization efforts, quality management systems and emerging challenges affecting pharmaceutical and healthcare regulation in the region.
The summit is widely recognized as a strategic meeting place for regulatory affairs specialists and industry stakeholders to advance understanding of evolving GCC pharmaceutical regulations, share updates on compliance requirements and foster collaboration among regulators and the life sciences sector across Gulf markets.
Regulatory Modernization and Trends in the GCC
The Gulf Cooperation Council (GCC), consisting of Bahrain, Kuwait, Oman, Qatar, Saudi Arabia and the United Arab Emirates (UAE), has a recognized regulatory landscape with individual national authorities overseeing pharmaceutical and healthcare market product authorization, drug registration processes, safety and quality across these member states. Regulatory agencies in the region include Saudi Arabia’s Saudi Food and Drug Authority (SFDA) and the UAE’s Ministry of Health and Prevention (MOHAP), among others, each operating distinct framework for GCC market authorization, pharmacovigilance system and compliance.
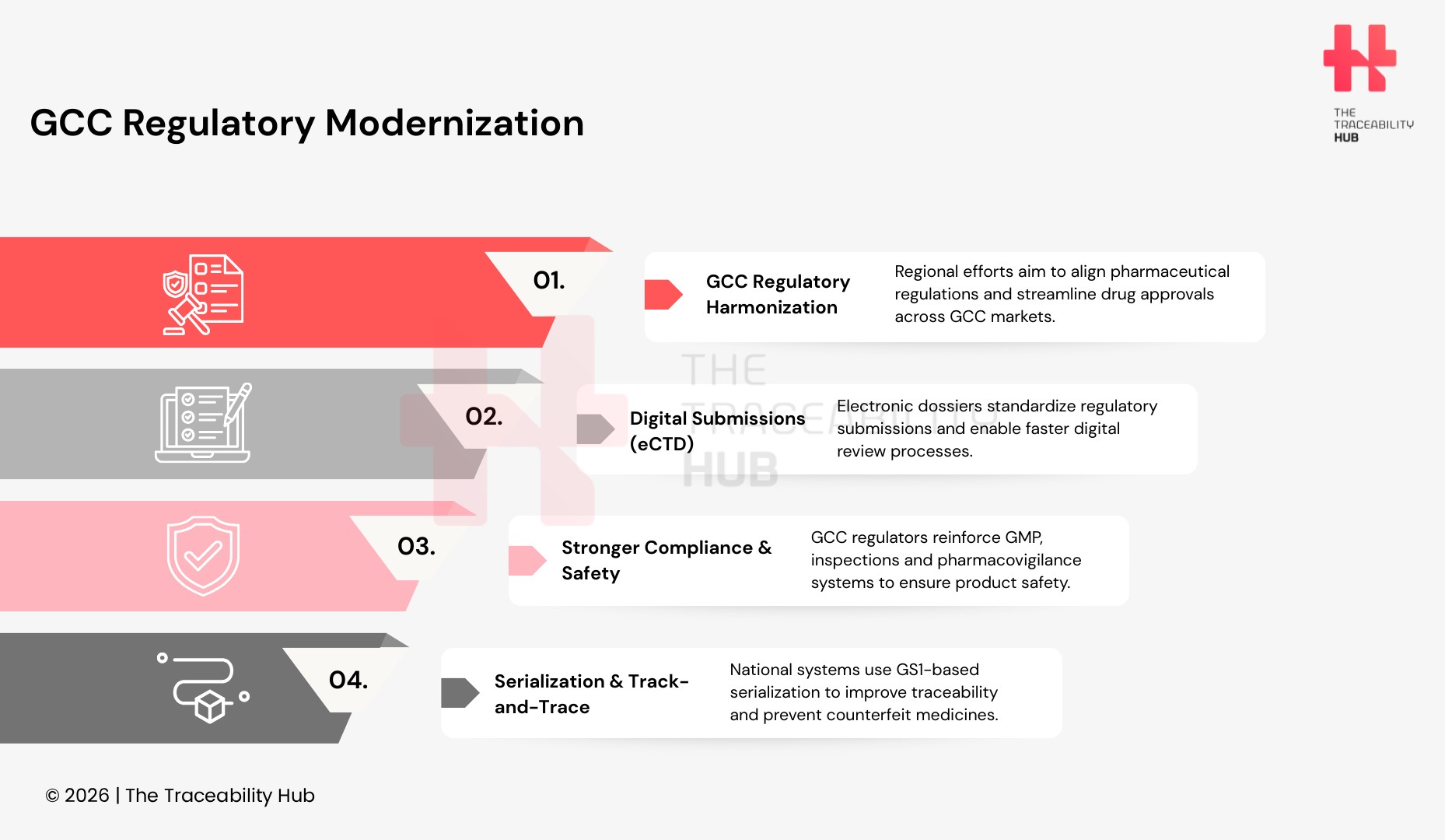
Regulatory evolution in the GCC reflects ongoing efforts toward harmonizing technical dossier and review requirements, including adoption of Common Technical Document (CTD) formats, GCC eCTD electronic submissions and reliance pathways that recognize approvals from established regulatory agencies like the US FDA and European regulators. This regulatory harmonization aims to streamline review processes and reduce duplication; albeit full convergence remains a longer-term objective.
Key trends shaping the GCC regulatory environment in 2025-2026 include:
- Continued collaboration on harmonization of regulatory requirements across GCC authorities, including streamlined dossier structures and shared risk-based evaluation principles aligned with international regulatory standards.
- Increased use of electronic submission specifications such as the eCTD framework adapted for GCC region filings, facilitating digital review workflows.
- Growing emphasis on pharmacovigilance and post-market safety systems as part of GCC regulatory expectations in individual GCC states.
- Advancements in pharmaceutical traceability and digital compliance infrastructure, and track-and-trace systems across GCC supply chains, including adoption of GS1 standards for barcode identification and serialization by several member states.
- Expansion of regulatory engagement with innovative therapies and biosimilars, supported by evolving GCC regulatory frameworks and alignment with international guidance on safety, quality and efficacy.
While each GCC country maintains its own regulatory authority and processes, the collective movement toward regulatory harmonization, digitalization and enhanced safety surveillance highlights the region’s ongoing regulatory modernization and its implications for market access, compliance operations and public health outcomes.
Key Themes Expected at the GCC Regulatory Affairs Summit 2026
Regulatory Harmonization & Regional Alignment
The GCC Regulatory Affairs Summit 2026 addresses regulatory updates and policy developments impacting pharmaceutical and healthcare markets across the Gulf Cooperation Council (GCC) member states. Regulatory harmonization remains an ongoing objective within the region, particularly through initiatives such as the GCC Central Committee for Drug Registration (GCC-DR), which aims to facilitate cooperation and alignment among national authorities. Discussions at the summit focus on GCC regulatory requirements, regional developments and cross-jurisdictional considerations relevant to companies operating in multiple GCC markets.
Digitalization of Regulatory Processes
Digital transformation in regulatory affairs is a documented trend across GCC regulatory authorities. Several member states, including Saudi Arabia through the Saudi Food and Drug Authority (SFDA), have implemented mandatory electronic submission requirements based on the electronic Common Technical Document (eCTD) format for pharmaceutical product registration. GCC Module 1 specifications define regional requirements for structured electronic submissions, supporting standardized dossier formats, metadata-driven documentation, and digital review processes. These developments reflect the broader adoption of electronic regulatory systems and digital compliance infrastructures across the region.
Compliance, Quality Systems & Inspection Readiness
The summit program includes components such as the GCC Quality Conference, which focuses on Good Manufacturing Practice (GMP), quality systems, and regulatory compliance. GCC regulatory authorities maintain inspection frameworks aligned with internationally recognized GMP standards and risk-based audit methodologies. As regulatory oversight continues to evolve, inspection preparedness, quality management systems and data integrity remain important areas of focus for pharmaceutical manufacturers operating within GCC regulatory jurisdictions.
Market Access & Accelerated Approval Pathways
Market access considerations are central to regulatory affairs activities in the GCC pharmaceutical sector. National authorities such as the SFDA and UAE Ministry of Health and Prevention (MOHAP) have established regulatory pathways governing product registration, electronic dossier submission, lifecycle management, and post-approval requirements. While timelines and approval mechanisms vary by country, regulatory updates and approval frameworks form part of the broader strategic discussions addressed at regional regulatory forums such as this summit.
Supply Chain Integrity & Traceability
Supply chain integrity, pharmaceutical serialization, and product traceability have become increasingly relevant within GCC regulatory modernization efforts. Several member states have introduced track-and-trace systems, barcode verification mechanisms, and serialization requirements for pharmaceutical products to combat counterfeiting and enhance supply chain visibility. These regulatory measures align with global GS1 standards for product identification and authentication and are part of the broader compliance landscape impacting pharmaceutical stakeholders across GCC markets.
GCC Regulatory Modernization
Why This Matters for Traceability and Digital Compliance
For The Traceability Hub audience, the GCC regulatory landscape is increasingly shaped by digital submission standards and supply chain control systems that rely on high-quality, interoperable master data. On the submissions side, GCC regulators have formalized requirements for electronic dossiers through the GCC eCTD Module 1 specifications, which standardize how regional administrative information is structured, validated and exchanged in the electronic Common Technical Document (eCTD) format.
On the supply chain side, interoperable master traceability is not theoretical: it is being operationalized through national track-and-trace programs. In Saudi Arabia, SFDA’s Drug Track and Trace System (RSD) requires unique serialization to help prevent counterfeit medicines, ensure regulatory traceability compliance, and strengthen end-to-end visibility. In the UAE, MOHAP’s Tatmeen is a national track-and-trace platform designed to enable pharmaceutical traceability using GS1-aligned serialization, supporting anti-counterfeiting controls and enabling functions such as recalls efficiency and broader supply chain visibility.
In parallel, regulators are reinforcing post-market safety expectations through formal pharmacovigilance frameworks (e.g., SFDA’s guidance on Good Pharmacovigilance Practices), which increases the value of reliable product identifiers and structured data flows across the product lifecycle. Practically, this is where pharmaceutical traceability becomes a digital compliance enabler: robust serialization and product identity data can support faster, more targeted recalls, strengthen anti-counterfeiting safeguards, and improve inspection readiness by making provenance and distribution data easier to evidence.
Finally, implementing these GCC regulatory requirements typically demands tighter coordination between regulatory affairs, quality, IT governance, and pharmaceutical supply chain teams, because compliance is increasingly dependent on interoperable systems, master data governance, and consistent cross-border reporting standards.
A Regional Platform with Global Relevance
The Gulf Cooperation Council (GCC) is geographically positioned at the intersection of Europe, Asia and Africa, making it a significant commercial and pharmaceutical logistics hub for global healthcare supply chains. Each GCC member state maintains its own regulatory authority and national framework governing product registration, labeling, pharmacovigilance and post-market surveillance. As a result, companies operating across multiple GCC jurisdictions must comply with country-specific electronic submission procedures, labeling standards requirements, safety reporting obligations and lifecycle management rules issued by national regulators such as the Saudi Food and Drug Authority (SFDA) and the UAE Ministry of Health and Prevention (MOHAP).
In this GCC regulatory environment, regional industry forums — including the GCC Regulatory Affairs Summit 2026 — provide structured opportunities for regulatory professionals, pharmaceutical compliance leaders and digital industry representatives and solution providers to exchange information on regulatory updates, compliance requirements and digital submission standards such as eCTD. By convening stakeholders across different GCC markets, these events support knowledge sharing and practical alignment on evolving regulatory expectations within the region.
The Strategic Takeaway
Regulatory authorities across the Gulf Cooperation Council (GCC) have implemented formal digital submission requirements, structured pharmacovigilance frameworks, and national track-and-trace systems that are reshaping regulatory compliance operations in the region. The adoption of the electronic Common Technical Document (eCTD) format standardizes electronic regulatory submissions across member states. In parallel, authorities such as the Saudi Food and Drug Authority (SFDA) and the UAE Ministry of Health and Prevention (MOHAP) have established structured pharmacovigilance guidance and national pharmaceutical track-and-trace platforms, reinforcing data integrity, product identification controls, anti-counterfeiting safeguards and supply chain oversight requirements.
Within this GCC regulatory framework, compliance activities increasingly depend on structured electronic data, serialized product identifiers, and interoperable reporting systems. For pharmaceutical, medical device and regulated healthcare product stakeholders, forums such as the GCC Regulatory Affairs Summit 2026 provide updates on regulatory requirements, digital submission standards and quality expectations currently applied across GCC markets. As regulatory oversight incorporates electronic submissions, post-market safety monitoring and serialization programs, pharmaceutical traceability systems form part of the documented compliance infrastructure governing healthcare products in the GCC region.
Read more: RFID Retail Systems: The Backbone of Data-Driven Shopping Success






