Familiar Signals, Misleading Assumptions
As organizations begin planning for the FDA’s transition to the uniform 12-digit NDC format (NDC12 transition, FDA NDC format update), many are comparing it to DSCSA (Drug Supply Chain Security Act compliance).
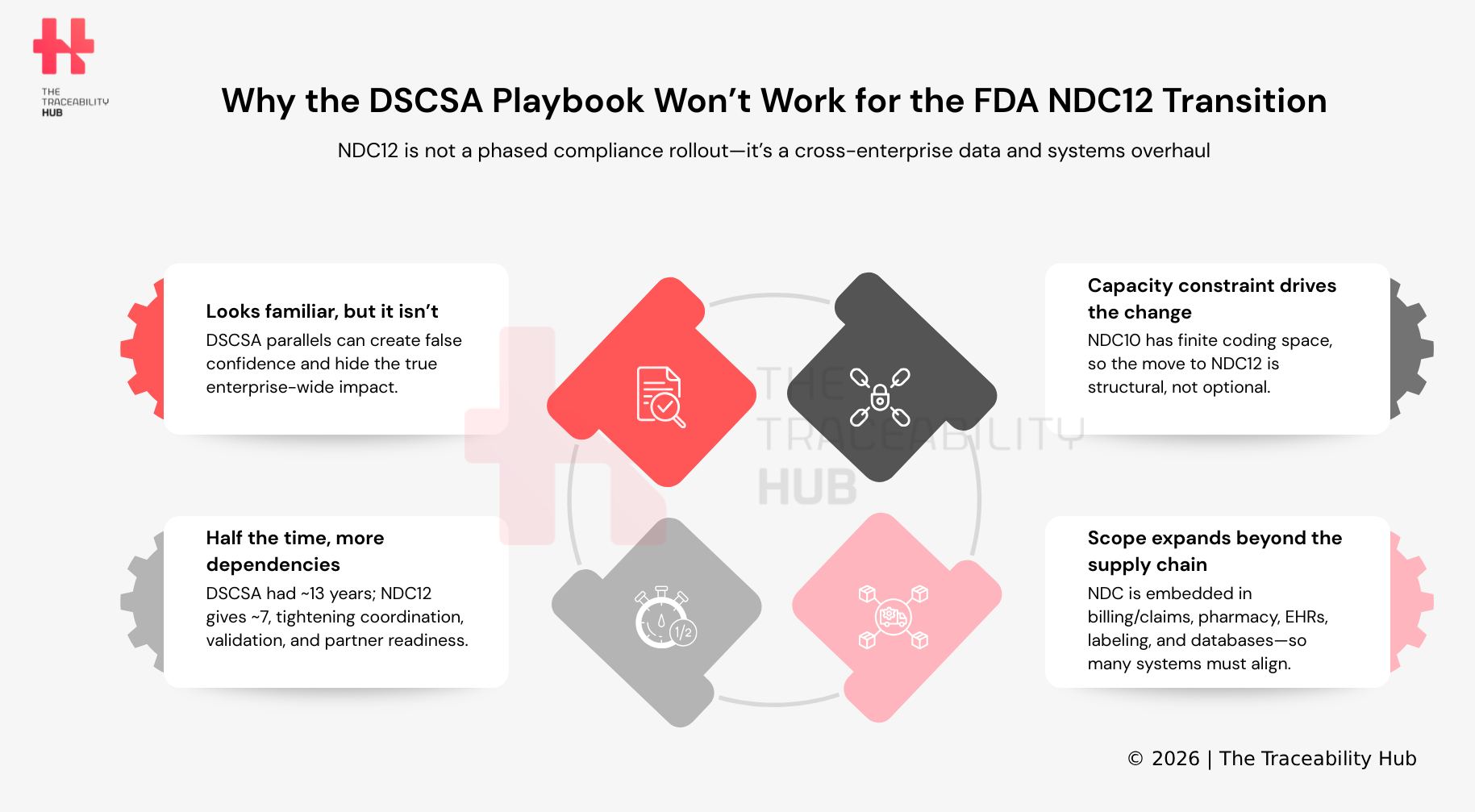
The comparison is understandable. DSCSA was one of the most significant recent regulatory changes affecting the U.S. pharmaceutical supply chain, and its requirements were phased in over several years, demanding substantial coordination and operational change across trading partners (supply chain partners and stakeholders).
However, that familiarity can create false confidence – a common regulatory compliance risk. When a new challenge appears similar to a previous one, it is easy to assume the same approach will apply. In the case of the 12-digit NDC transition, that assumption may lead organizations to underestimate both the breadth of the enterprise-wide system impact and the execution conditions (implementation complexity and constraints) that will shape implementation.
Structural Constraint, Not Just Regulatory Change
The Drug Supply Chain Security Act (DSCSA) was driven by pharmaceutical compliance regulations. The requirements were significant, but implementation was phased over time, and timelines evolved as the industry worked through operational challenges (compliance timelines and phased rollout).
The transition to the FDA’s uniform 12-digit NDC format is driven by a different underlying constraint (data structure limitation). The current 10-digit NDC structure has finite capacity, particularly in the availability of labeler codes – NDC code limitations. While that capacity has not yet been exhausted, it is limited, and the transition is intended to ensure sufficient coding space for future drug products or future-proofing drug identification systems.
This distinction changes the nature of the transition. Regulatory timelines can evolve as implementation challenges emerge. Capacity constraints, however, are less flexible and require proactive data architecture transformation. Organizations that expect the same level of timing flexibility seen during DSCSA implementation may find that the conditions surrounding the 12-digit NDC transition are more constrained.
Expanding Scope Across the Healthcare Ecosystem
The Drug Supply Chain Security Act (DSCSA) was a major undertaking, but it was primarily focused on the U.S. prescription drug supply chain, pharmaceutical distribution network and its trading partners.
The impact of the FDA’s transition to a uniform 12-digit National Drug Code (NDC) format reaches well beyond that boundary. The National Drug Code is used across both prescription and over-the-counter products and is embedded across a wide range of healthcare IT systems, including billing and claims processing, pharmacy systems, electronic health records (EHR systems), barcoding and labeling systems, and regulatory databases.
Consequently, transition influences not only supply chain operational procedures but also establishes a uniform format for product identifiers (standardized drug identification). It alters how product data is organized and transmitted across various systems and environments. The breadth of impact is both wider and more dispersed, reflecting increased digital health ecosystem integration.
Seven Years vs Thirteen Changes the Equation
At first glance, a seven-year NDC12 implementation timeline may seem reasonable. However, when considered in context, it is less so. DSCSA commenced in 2013; following extensions and enforcement discretion, the industry had approximately thirteen years to achieve a full DSCSA compliance timeline. In contrast, NDC12 allows for only about seven years.
Consequently, the industry has roughly half the preparation time for a transformation that not only impacts the pharmaceutical supply chain system but also extends into a wider healthcare ecosystem and healthcare infrastructure. This intensifies the urgency for organizations to evaluate system implications, standardize data frameworks, cross-functional and partner collaboration, drug labeling updates, and complete validation processes across numerous environments. These tasks are interdependent and compete for limited resources.
The primary challenge will not be awareness, but rather the capacity to execute these complex requirements.
Why the DSCSA Playbook Breaks Down
The experience with the Drug Supply Chain Security Act (DSCSA) provides meaningful insights – lessons learned in pharma compliance. It underscores the significance of proactive planning, effective cross-functional collaboration, and strong alignment among trading partners. Nonetheless, it is important to recognize that direct comparisons have certain limitations.
DSCSA primarily tested the U.S. pharmaceutical supply chain. The transition to the FDA’s uniform 12-digit NDC format has implications that extend beyond that boundary, impacting systems across the broader healthcare data infrastructure and ecosystem. The National Drug Code (NDC) plays a central role in how products are identified and how related data is structured and linked across systems (master data management and product identification standards).
Assuming uniform scope, timing, or flexibility can lead to significant compliance risk and implementation gaps. Issues that seem routine may, in fact, be more complex and interconnected than initially perceived. It is crucial to acknowledge these distinctions to ensure effective regulatory strategy, transformation planning and decision-making.
Why the DSCSA Playbook Won’t Work for the FDA NDC12 Transition
Start With the Right Questions
Organizations preparing for the transition to the FDA’s uniform 12-digit NDC format typically assess their starting point carefully. They review how the National Drug Code (NDC) is utilized throughout their operations (NDC usage across systems), identify which systems and partners present the most significant system dependencies and integration points, and pinpoint areas where constraints may arise during implementation.
This method shifts the emphasis from mere compliance to comprehensive coordination, addressing the interconnected requirements among systems and stakeholders. Such an approach more effectively reflects the cross-functional complexity of this change (organizational and technical complexity in NDC12 transition).
Coming soon: Time Out of Refrigeration (TOR): A Critical Challenge in the Era of RNA and Biologic Therapies






