FDA Finalizes NDC12: A New Standard for Drug Identification
The FDA, Food and Drug Administration, the U.S. federal agency responsible for protecting public health by ensuring the safety, efficacy, and security of food, drugs, medical devices, and other health-related products, has officially finalized the transition to the 12-digit National Drug Code (NDC12), marking a significant milestone for the pharmaceutical industry and healthcare data standards.
For many stakeholders, this moment has been anticipated for years. The capacity limits of the current 10-digit NDC structure (legacy drug identification system), have long been recognized and widely discussed across regulatory forums and industry working groups.
Now, the transition is no longer theoretical: it is official (National Drug Code Format | FDA).
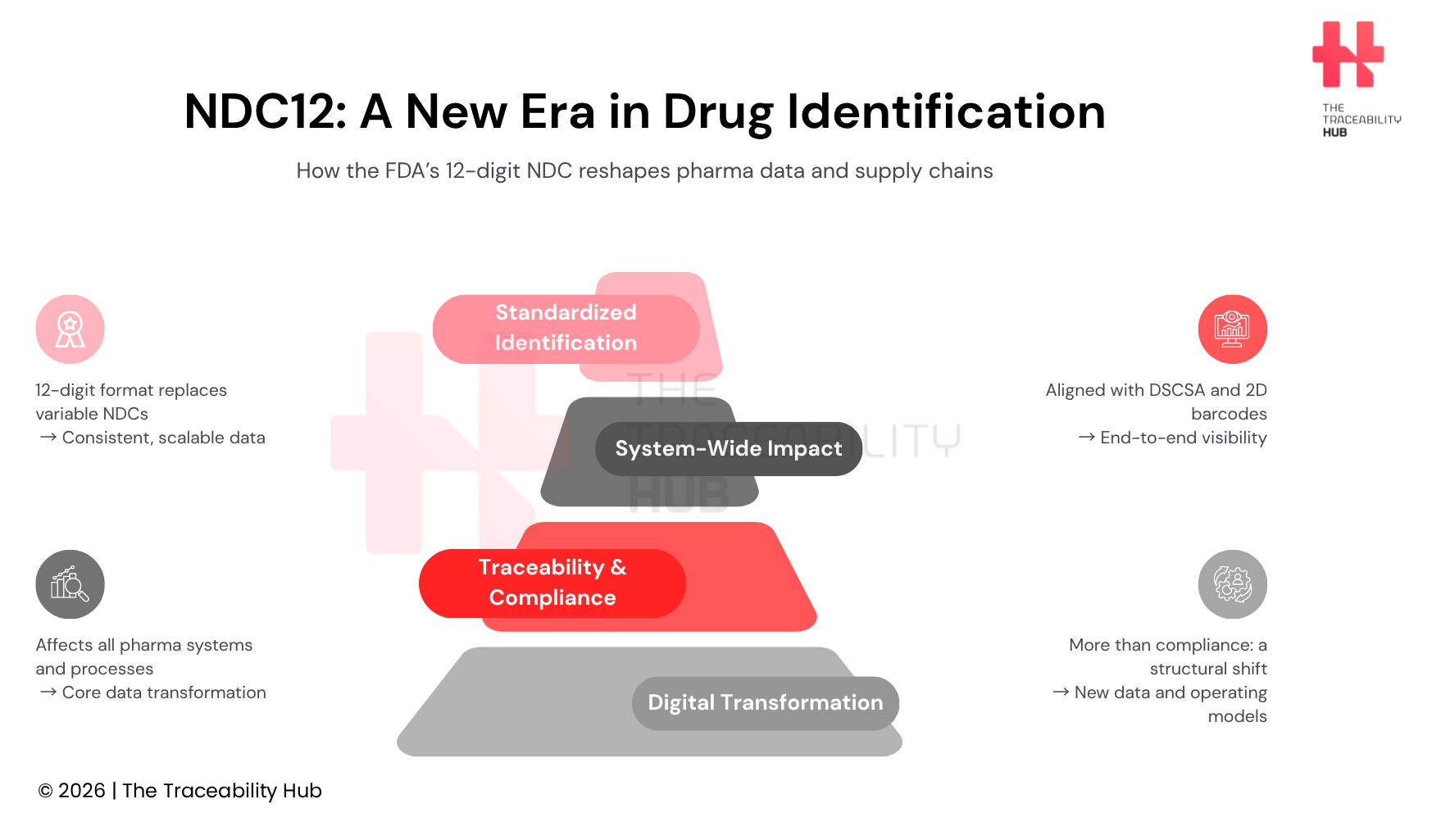
Under the final rule, “Revising the National Drug Code Format and Drug Label Barcode Requirements”, the FDA adopts a uniform 12-digit NDC format, the standard used to uniquely identify drugs marketed in the United States. Currently, NDCs are assigned as 10-digit numbers in multiple configurations (variable NDC formats). This variability will be replaced by a single, standardized format, improving data consistency and interoperability across healthcare systems.
The rule will take effect on March 7, 2033. On that date, the FDA will begin assigning new 12-digit NDCs and will automatically convert all existing 10-digit codes into the new format. The change applies broadly to human drugs, including biological products, as well as animal drugs, reinforcing a unified drug identification and regulatory compliance framework.
A seven-year transition period, running from March 5, 2026 to March 6, 2033, has been established to allow manufacturers, distributors, repackagers, pharmacies, healthcare providers, payors, and other pharmaceutical supply chain stakeholders to prepare. During this time, the FDA will continue assigning 10-digit NDCs, while encouraging all affected parties to update systems, processes, and infrastructure to support the future 12-digit NDC standard and healthcare data interoperability requirements.
Beyond the Format: Why NDC12 Impacts the Entire Healthcare Ecosystem
At first glance, the change appears straightforward: the National Drug Code expands from ten digits to a standardized twelve-digit format.
But the operational implications are much larger than the number of digits might suggest, especially in the context of pharmaceutical supply chain digitization and healthcare system integration.
The NDC sits at the center of the healthcare ecosystem. It is the reference point used to identify pharmaceutical products across a wide range of interconnected environments: manufacturing execution systems (MES), enterprise resource planning (EPR) platforms, wholesale and distribution networks, pharmacy management systems, reimbursement and billing infrastructures, as well as regulatory reporting databases such as FDA drug listing system.
Because of this, the NDC is not just a regulatory identifier – it is a core master data element in pharmaceutical data governance embedded in virtually every transaction involving a drug product. It enables product recognition, data exchange, traceability, and financial flows across the entire lifecycle of a medicine.
Changing that identifier, therefore, does not simply affect regulatory filings or labeling documentation. It has direct consequences on system interoperability, master data management, and supply chain operations, including how systems communicate with each other, how product master data is structured and maintained, how barcodes are generated and interpreted, and how transactions are processed across the supply chain.
For example, systems will need to be capable of handling both 10-digit and 12-digit NDC formats (dual-format data handling) during the transition period from March 7, 2033 to March 6, 2036. At the same time, companies will be required to update drug labeling and barcode systems by introducing the 12-digit format – typically by adding leading zeros to existing segments – while ensuring continuity in operations and avoiding disruptions in distribution or dispensing.
The impact also extends to barcode requirements and drug serialization systems. Under the new rule, a 2D data matrix barcode compliant with recognized standards such as GS1 can encode the 12-digit NDC alongside other critical data elements, including serial number, lot number, and expiration date. This creates the opportunity to align NDC identification more closely with Drug Supply Chain Security Act (DSCSA) requirements, potentially enabling a single barcode to support pharmaceutical traceability, serialization, and regulatory compliance.
DSCSA, enacted on November 27, 2013, is a federal law that establishes an electronic, interoperable system to trace prescription drugs through the U.S. supply chain, enhancing safety and preventing counterfeit or harmful drugs from reaching patients.
NDC12: A New Era in Drug Identification
From Compliance to Transformation: What NDC12 Really Means
This is why many organizations are beginning to frame the transition in a different way.
Not as a compliance exercise.
But as a pharmaceutical digital transformation.
What initially appears as a format update is, in reality, a structural change that touches data governance, system interoperability, labeling strategies, and supply chain operations. It requires coordination across multiple functions – regulatory, IT, manufacturing, quality, and commercial – and across multiple external partners within the healthcare ecosystem.
Major industry shifts rarely occur all at once. They typically begin with a triggering event – often a regulatory change or a structural limitation – and then expand outward, influencing every layer of the ecosystem that depends on that foundation.
With the finalization of NDC12, that triggering moment has arrived signaling a broader shift toward standardized data, interoperability, and supply chain modernization in pharma.
Industry Response and What Comes Next
The next opportunity for industry leaders to discuss the implications of this shift will take place at the upcoming Distribution Management Conference (DMC), hosted by the Healthcare Distribution Alliance (HDA). For the first time since the rule was finalized, manufacturers, distributors, technology providers, and regulators will come together to exchange perspectives on what NDC12 implementation and pharmaceutical supply chain transformation will mean in practice.
Moments like this are particularly revealing. Industry events often serve as an early indicator of how organizations are interpreting and prioritizing a new regulatory requirement and data standardization initiative.
Are leaders already mobilizing resources and cross-functional teams?
Where do companies anticipate the greatest operational risks in NDC12 compliance and system upgrades?
Which parts of the business – IT systems, labeling, supply chain execution, or data governance – will feel the impact first?
The answers to these questions will begin to shape how quickly – and how effectively – the industry moves from awareness to execution in this pharmaceutical data transformation journey.
In the next article, we will share key observations emerging from DMC discussions, highlighting how different stakeholders are approaching the transition and where early patterns are starting to form.
Because while the rule may now be final, the real work is only just beginning.
NDC12 is not simply a format update. It represents the next enterprise-wide transformation in healthcare, driven by regulatory change, data standardization, and supply chain digitization.
Read more: Traceability in ASPICE Automotive Projects: ISO 26262 Compliance Best Practices






